Actualités
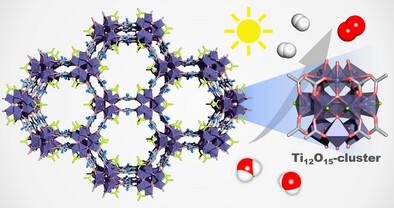
Voir toutes les actualitésA Novel Ti12-Based Metal-Organic Framework for Photocatalytic Hydrogen Evolution
Bio-inspired Electrocatalysts for Water Oxidation
From Automated High-Throughput Experimentation to Self-Driving Laboratories: The Swiss Cat⁺ Experience
Strategies for Selective Chemical Sensing and Imaging with Polymeric Nanospheres
A Quest for Bio-Orthogonal Transition Metal Catalysis in Living Cells
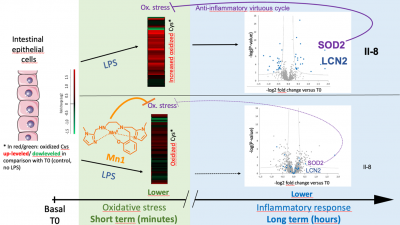
Kinetic Redox Shotgun Proteomics Reveals Specific Lipopolysaccharide Effects on Intestinal Epithelial Cells, Mitigated by a Mn Superoxide Dismutase Mimic
Recherche et formation
 |
 |
 |
 |
Suivez-nous
| Tweets by Chimie__ENS |