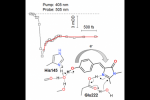
Because of growing applications in advanced fluorescence imaging, the mechanisms and dynamics of photoinduced reactions in reversibly photoswitchable fluorescent proteins are currently attracting much interest. We report the fi rst timeresolved study of the photoswitching of Dreiklang, so far the only fluorescent protein to undergo reversible photoinduced chromophore hydration. Using broadband femtosecond transient absorption spectroscopy, we show that the reaction is triggered by an ultrafast deprotonation of the chromophore phenol group in the excited state in 100 fs. This primary step is accompanied by coherent oscillations that we assign to its coupling with a low-frequency mode, possibly a deformation of the chromophore hydrogen bond network. A ground-state intermediate is formed in the picosecond− nanosecond regime that we tentatively assign to the deprotonated water adduct. We suggest that proton ejection from the phenol group leads to a charge transfer from the phenol to the imidazolinone ring, which triggers imidazolinone protonation by nearby Glu222 and catalyzes the addition of the water molecule.

Consultez le communiqué de presse associé à cet article : Comprendre le comportement d’un chromophore fluorescent !
References:
Photoinduced Chromophore Hydration in the Fluorescent Protein Dreiklang Is Triggered by Ultrafast Excited-State Proton Transfer Coupled to a Low-Frequency Vibration
Fabien Lacombat, Pascal Plaza, Marie-Aude Plamont, and Agathe Espagne
J. Phys. Chem. Lett. 2017, 8, 1489−1495
DOI: 10.1021/acs.jpclett.7b00348
Photoinduced Chromophore Hydration in the Fluorescent Protein Dreiklang Is Triggered by Ultrafast Excited-State Proton Transfer Coupled to a Low-Frequency Vibration